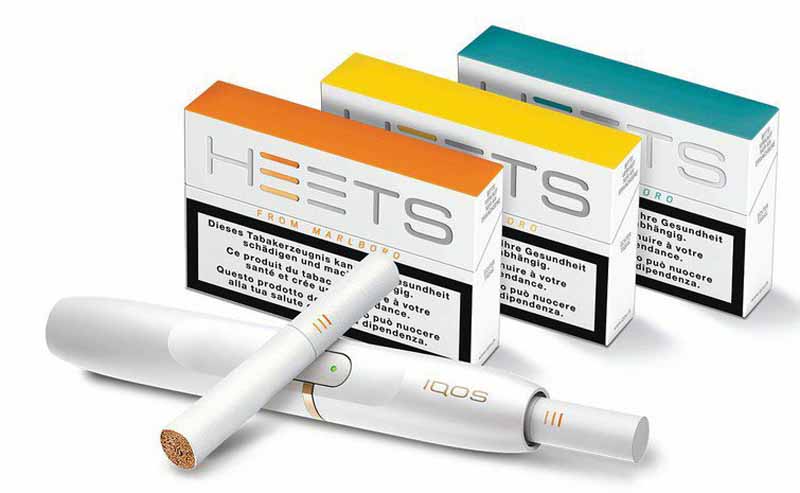
The U.S. trade representative will consider the FDA’s view while mulling an import ban.Read More
Tags :IQOS
The agency has instituted a ban preventing IQOS from being sold in the U.S. in 60 days.Read More
IQOS Iluma provides a cleaner, more consistent experience than previous versions, according to the company. Read More
CEO Olczak details next steps toward becoming a majority smoke-free business at Goldman Sachs Forum. Read More
If the initial findings are upheld, the commission could potentially ban IQOS imports into the U.S.Read More
The device features a longer battery life and quicker recharge than its predecessor. Read More
Atria wants the FDA's help in convincing Americans that nicotine isn't linked to cancer, a letter states.Read More
But FDA approval of new technology could speed up things, according to Goldman Sachs' Bonnie Herzog.Read More
The openings mark a significant step towards achieving the company's vision of a smoke-free future.Read More
The Therapeutic Goods Administration in Australia has rejected an application from Philip Morris that would have allowed the sale of heated-tobacco products.Read More